236
Polyamide-Polyamine Cryptand as a New Generation Dicarboxylate Receptor: Di-anion Binding Studies in the Solid State, in Solution, and in the Gas Phase
S. Chakraborty, S. Saha, L.M.P. Lima, U. Warzok , Sayan Sarkar, B. Akhuli, M. Nandi, S. Bej, N.N. Adarsh, C.A. Schalley, R. Delgado, P. Ghosh – 2017
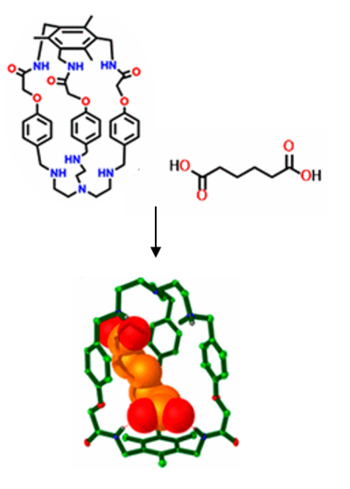
A polyamide-polyamine hybrid macrobicycle L is explored with respect to its ability to bind α,ω-dicarboxylate anions. Potentiometric studies of protonated L with the series of dianions from succinate (suc2–) through, glutarate (glu2–), alpha-ketoglutarate (kglu2–), adipate (adi2–), pimelate (pim2–), suberate (sub2–), to azelate (aze2–) have shown adipate preference with association constant value of K = 4900 in a H2O/DMSO (50:50 v/v) binary solvent mixture. The binding constant increases from glu2– to adi2– and then continuously decreases with the length of the anion chain. Further, potentiometric studies suggest that hydrogen bonding between the guest anions and the amide / ammonium protons of the receptor also contributes to the stability of the associations along with electrostatic interactions. Negative-mode electrospray ionization of aqueous solutions of host-guest complexes shows clear evidence for the selective formation of 1:1 complexes. Single crystal X-ray structures of the host-guest complexes of the receptor with glutaric acid, alpha-ketoglutaric acid, adipic acid, pimelic acid, suberic acid and azelaic acid assisted in the understanding of the observed binding preferences. Moreover, the crystal structure of the adipate complex shows shorter hydrogen bond distances of the encapsulated anion in the amide and ammonium cleft of the receptor compared to other guests. The solid-state structures reveal a size / shape complementarity between the host and the dicarboxylate anions, which is nicely reflected in the solution state binding studies. Thus, size complementarity between the receptor cavity and the guest dianions is another important factor determining the binding strength.
Polyamide-Polyamine Cryptand as a New Generation Dicarboxylate Receptor: Di-anion Binding Studies in the Solid State, in Solution, and in the Gas Phase