New Publication in Proteomics
Higher order structure of proteins
News from Feb 02, 2015
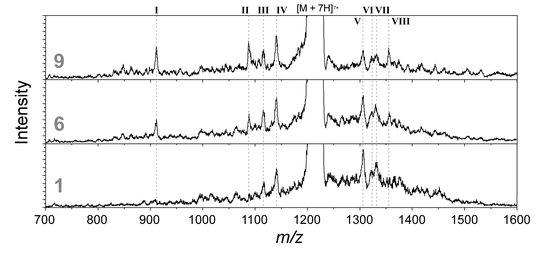
Analyzing the higher order structure of proteins with conformer-selective ultraviolet photodissociation
The top-down approach in protein sequencing requires simple methods in which the analyte can be readily dissociated at every position along the backbone. In this context, ultraviolet photodissociation (UVPD) recently emerged as a promising tool because, in contrast to slow heating techniques such as collision induced dissociation (CID), the absorption of UV light is followed by a rather statistically distributed cleavage of backbone bonds. As a result, nearly complete sequence coverage can be obtained. It is well-known, however, that gas-phase proteins can adopt a variety of different, sometimes coexisting conformations and the influence of this structural diversity on the UVPD fragmentation behavior is not clear. Using ion mobility-UVPD-mass spectrometry we recently showed that UVPD is sensitive to the higher order structure of gas-phase proteins. In particular, the cis/trans isomerization of certain proline peptide bonds was shown to significantly influence the UVPD fragmentation pattern of two extended conformers of 11+ ubiquitin. Building on these results, we here provide conformer-selective UVPD data for 7+ ubiquitin ions, which are known to be present in a much more diverse and wider ensemble of different structures, ranging from very compact to highly extended species. Our data show that certain conformers fall into groups with similar UVPD fragmentation pattern. Surprisingly, however, the conformers within each group can differ tremendously in their collision cross section. This indicates that the multiple coexisting conformations typically observed for 7+ ubiquitin are caused by a few, not easily inter-convertible, subpopulations.
go to the article: DOI 10.1002/pmic.201400480




