Molecular Recognition: Receptor Chemistry and Anion Binding
Molecular recognition is the basis for almost all other processes in supramolecular chemistry, for example template effects, that organize the components in a suitable manner to obtain a certain geometrical arrangement, or self-assembly and self-sorting, with which the complexes of interest can be programmed.
The Schalley group has examined receptors for neutral molecules such as caffeine, investigated the binding interactions in amide pseudorotaxanes in detail with respect to substituent effects, and studied anion binding by weak forces such as anion-pi interactions and C-H...anion interactions. Dispersive interactions have been studied together with our cooperation partners in Aachen (Markus Albrecht) and Jyväskylä/Finland (Kari Rissanen).
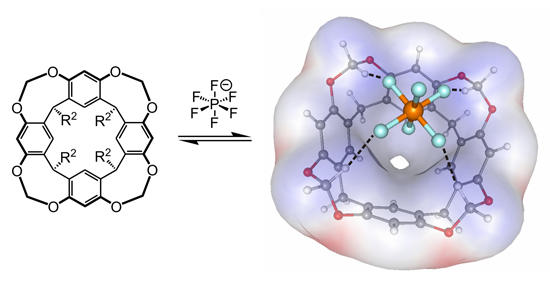
Resorcinarene cavitand binding hexafluorophosphate through C-H...anion interactions
Highlights
Dispersive Interactions
- Chasing weak forces: hierarchically assembled helicates as a probe for the evaluation of the energetics of weak interactions
D. Van Craen, W.H. Rath, M. Huth, L. Kemp, C. Räuber, J. Wollschläger, C.A. Schalley, A. Valkonen, K. Rissanen, M. Albrecht
J. Am. Chem. Soc. 2017, 139, 16959-16966
Anion Binding & Anion-pi Interactions
- Experimental evidence for the functional relevance of anion-pi interactions
R.E. Dawson, A. Hennig, D.P. Weimann, D. Emery, V. Ravikumar, J. Montenegro, T. Takeuchi, S. Gabutti, M. Mayor, J. Mareda, C.A. Schalley, S. Matile
Nature Chem. 2010, 2, 533-538
- Anion Binding to Resorcinarene-Based Cavitands: The Importance of C-H...Anion Interactions
S.S. Zhu, H. Staats, K. Brandhorst, J. Grunenberg, F. Gruppi, E. Dalcanale, A. Lützen, K. Rissanen, C.A. Schalley
Angew. Chem. 2008, 120, 800-804; Angew. Chem. Int. Ed. 2008, 47, 788-792
Amide Pseudorotaxanes
- Substituent Effects on Axle Binding in Amide Pseudorotaxanes: Comparison of NMR Titration and ITC Data with DFT Calculations
L. Kaufmann, E.V. Dzyuba, F. Malberg, N.L. Löw, M. Groschke, B. Brusilowskij, J. Huuskonen, K. Rissanen, B. Kirchner, C.A. Schalley
Org. Biomol. Chem. 2012, 10, 5954-5964
- CH•••O Hydrogen Bonds in "Clicked" Diketopiperazine-Based Amide Rotaxanes
E.V. Dzyuba, L. Kaufmann, N.L. Löw, A.K. Meyer, H.D.F. Winkler, K. Rissanen, C.A. Schalley
Org. Lett. 2011, 13, 4838-4841
- Uncovering Individual Hydrogen Bonds in Rotaxanes by Frequency Shifts
B. Kirchner, C. Spickermann, W. Reckien, C.A. Schalley
J. Am. Chem. Soc. 2010, 132, 484-494
- How Can Rotaxanes be Modified by Varying Functional Groups at the Axle? A Combined Theoretical and Experimental Analysis of Thermochemistry and Electronic Effects
C. Spickermann, T. Felder, C.A. Schalley, B. Kirchner
Chem. Eur. J. 2008, 14, 1216-1227
Caffeine Receptor
- First Artificial Receptor for Caffeine - A New Concept for the Complexation of Alkylated Oxopurines
S. R. Waldvogel, R. Fröhlich, C. A. Schalley
Angew. Chem. 2000, 112, 2580-2584; Angew. Chem. Int. Ed. 2000, 39, 2472-2475
