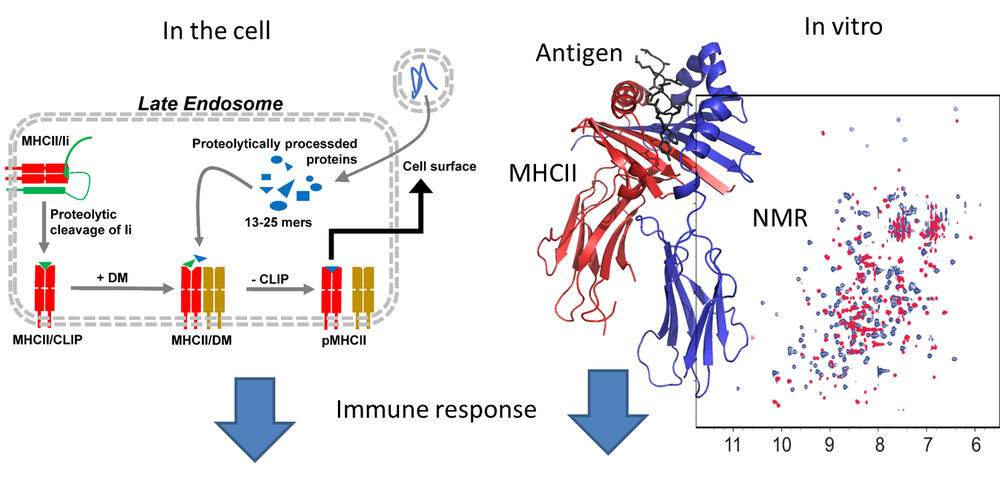
Our group is interested in the mechanisms and molecular logic that govern the molecular interactions of proteins. The focus is on proteins with a known functional impact on adaptive immunity or neuronal function. NMR spectroscopy is used to question the structural properties of molecules while mass spectrometric studies report on the compositional dynamics of proteomes when cells are challenged by intracellular or extracellular cues. A particular focus is on proteins of the human leukocyte antigen (HLA) family that present processed protein antigens to T cells. Here certain determinants of antigen processing and presentation are interrogated in the context of autoimmunity or viral infections. Activation of T cells by antigenic stimuli is followed by our group for proteins that modulate critical intracellular pathways. This ranges from crucial factors of T cell motility (ADAP) to modulators of membrane anchorage (palmitoyl transferases) and proteins that impact mRNA splicing (CD2BP2). In the case of proteins of the nervous system we are interested in factors that enable fast exocytosis or that play a role in the synaptic vesicle cycle (Syntaxin-1B, intersectin). In all cases, collaborations with groups interested in the biology of these molecules help to draw a more complete picture about their specific role in the cell and/or in the context of disease.
Keywords
- Freund
- NMR
- Protein Biochemistry
- Protein Interactions