Cryogenic Infrared Spectroscopy Reveals Structural Modularity in the Vibrational Fingerprints of Heparan Sulfate Diastereomers
Lettow, M.; Grabarics, M.; Greis, K.; Mucha, E.; Thomas, D. A.; Chopra, P.; Boons, G.-J.; Karlsson, R.; Turnbull, J. E.; Meijer, G.; Miller, R.L., von Helden, G.; Pagel, K.;* – 2020
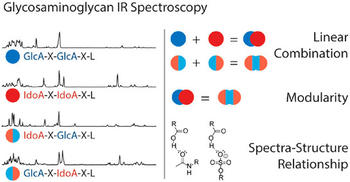
Heparan sulfate and heparin are highly acidic polysaccharides with a linear sequence, consisting of alternating glucosamine and hexuronic acid building blocks. The identity of hexuronic acid units shows a variability along their sequence, as d-glucuronic acid and its C5 epimer, l-iduronic acid, can both occur. The resulting backbone diversity represents a major challenge for an unambiguous structural assignment by mass spectrometry-based techniques. Here, we employ cryogenic infrared spectroscopy on mass-selected ions to overcome this challenge and distinguish isomeric heparan sulfate tetrasaccharides that differ only in the configuration of their hexuronic acid building blocks. High-resolution infrared spectra of a systematic set of synthetic heparan sulfate stereoisomers were recorded in the fingerprint region from 1000 to 1800 cm–1. The experiments reveal a characteristic combination of spectral features for each of the four diastereomers studied and imply structural modularity in the vibrational fingerprints. Strong spectrum-structure correlations were found and rationalized by state-of-the-art quantum chemical calculations. The findings demonstrate the potential of cryogenic infrared spectroscopy to extend the mass spectrometry-based toolkit for the sequencing of heparan sulfate and structurally related biomolecules.