Remote Participation during Glycosylation Reactions of Galactose Building Blocks: Direct Evidence from Cryogenic Vibrational Spectroscopy
Marianski, M.;* Greis, K.; Mucha, E.; Moon, S.; Pardo, A.; Kirschbaum, C.; Thomas, D.A.; Meijer, G.; von Helden, G.; Gilmore, K.; Seeberger, P.H.; Pagel, K.* – 2020
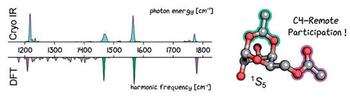
The stereoselective formation of 1,2‐cis‐glycosidic bonds is challenging. However, 1,2‐cis‐selectivity can be induced by remote participation of C4 or C6 ester groups. Reactions involving remote participation are believed to proceed via a key ionic intermediate, the glycosyl cation. Although mechanistic pathways were postulated many years ago, the structure of the reaction intermediates remained elusive owing to their short‐lived nature. Herein, we unravel the structure of glycosyl cations involved in remote participation reactions via cryogenic vibrational spectroscopy and first principles theory. Acetyl groups at C4 ensure α‐selective galactosylations by forming a covalent bond to the anomeric carbon in dioxolenium‐type ions. Unexpectedly, also benzyl ether protecting groups can engage in remote participation and promote the stereoselective formation of 1,2‐cis‐glycosidic bonds.