A Switchable synchronisation of pirouetting motions in a redox-active [3]rotaxane
Schröder, H. V.; Mekic, A.; Hupatz, H.; Sobottka, S.; Witte, F.; Urner, L. H.; Gaedke, M.; Pagel, K.; Sarkar, B.; Paulus, B.; Schalley, C. A.* – 2018
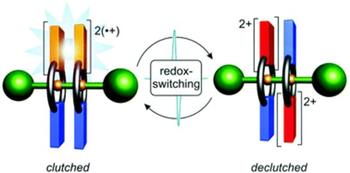
In this study, the crown/ammonium [3]rotaxane R2 is reported which allows a switchable synchronisation of wheel pirouetting motions. The rotaxane is composed of a dumbbell-shaped axle molecule with two mechanically interlocked macrocycles which are decorated with a redox-active tetrathiafulvalene (TTF) unit. Electrochemical, spectroscopic, and electron paramagnetic resonance experiments reveal that rotaxane R2 can be reversibly switched between four stable oxidation states (R2, R2˙+, R22(˙+), and R24+). The oxidations enable non-covalent, cofacial interactions between the TTF units in each state—including a stabilised mixed-valence (TTF2)˙+ and a radical-cation (TTF˙+)2 dimer interaction—which dictate a syn (R2, R2˙+, and R22(˙+)) or anti (R24+) ground state co-conformation of the wheels in the rotaxane. Furthermore, the strength of these wheel–wheel interactions varies with the oxidation state, and thus electrochemical switching allows a controllable synchronisation of the wheels’ pirouetting motions. DFT calculations explore the potential energy surface of the counter-rotation of the two interacting wheels in all oxidation states. The controlled coupling of pirouetting motions in rotaxanes can lead to novel molecular gearing systems which transmit rotational motion by switchable non-covalent interactions.