New Publication in JACS
Photodissociation of conformer selected ubiquitin
News from Jul 01, 2014
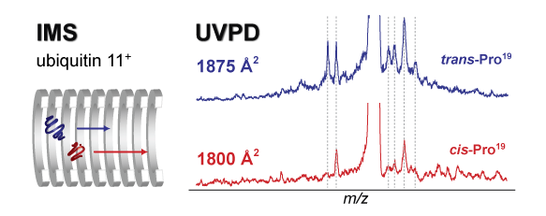
Photodissociation of Conformer-Selected Ubiquitin Ions Reveals Site-Specific cis/trans Isomerization of Proline Peptide Bonds
Ultraviolet photodissociation of gas-phase proteins (UVPD) has attracted increased attention in recent years. This growing interest is largely based on the fact that – in contrast to slow heating techniques such as collision induced dissociation (CID) - the cleavage propensity after absorption of UV light is distributed over the entire protein sequence, which can lead to a very high sequence coverage as required in typical top-down proteomics applications. However, in the gas phase proteins can adopt a multitude of distinct and sometimes coexisting conformations and it is not clear how this three-dimensional structure affects the UVPD fragmentation behavior. Using ion mobility – UVPD – mass spectrometry in conjunction with molecular dynamics simulations we provide the first experimental evidence that UVPD is sensitive to the higher order structure of gas-phase proteins. Distinct UVPD spectra were obtained for different extended conformations of 11+ ubiquitin ions. Assignment of the fragments showed, that the majority of differences arise from cis/trans isomerization of one particular proline peptide bond. Seen from a broader perspective these data highlight the potential of UVPD to be used for the structural analysis of proteins in the gas phase.
go to article doi: 10.1021/ja502994b




