Identification of an iridium-containing compound with a formal oxidation state of IX
Guanjun Wang, Mingfei Zhou, James T. Goettel, Gary J. Schrobilgen, Jing Su, Jun Li, Tobias Schlöder, Sebastian Riedel – 2014
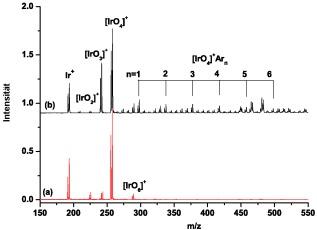
One of the most important classifications in chemistry and within the periodic table is the concept of formal oxidation states. The preparation and characterization of compounds containing elements with unusual oxidation states is of great interest to chemists. The highest experimentally known formal oxidation state of any chemical element is at present VIII, although higher oxidation states have been postulated. Compounds with oxidation state VIII include several xenon compounds (for example XeO₄ and XeO₃F₂) and the well-characterized species RuO₄ and OsO₄. Iridium, which has nine valence electrons, is predicted to have the greatest chance of being oxidized beyond the VIII oxidation state. In recent matrix-isolation experiments, the IrO₄ molecule was characterized as an isolated molecule in rare-gas matrices. The valence electron configuration of iridium in IrO₄ is 5d¹, with a formal oxidation state of VIII. Removal of the remaining d electron from IrO₄ would lead to the iridium tetroxide cation ([IrO₄]⁺), which was recently predicted to be stable and in which iridium is in a formal oxidation state of IX. There has been some speculation about the formation of [IrO₄]⁺ species, but these experimental observations have not been structurally confirmed. Here we report the formation of [IrO₄]⁺ and its identification by infrared photodissociation spectroscopy. Quantum-chemical calculations were carried out at the highest level of theory that is available today, and predict that the iridium tetroxide cation, with a Td-symmetrical structure and a d⁰ electron configuration, is the most stable of all possible [IrO₄]⁺ isomers.