Matrix Infrared Spectroscopy and Quantum-Chemical Calculations for the Coinage-Metal Fluorides: Comparisons of Ar-AuF, Ne-AuF, and Molecules MF2 and MF3
Xuefeng Wang, Lester Andrews, Felix Brosi, Sebastian Riedel – 2013
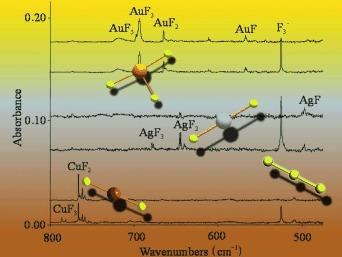
The reactions of laser-ablated Au, Ag, and Cu atoms with F₂ in excess argon and neon gave new absorptions in the MF stretching region of their IR spectra, which were assigned to metal-fluoride species. For gold, a NgAuF bond was identified in mixed neon/argon samples. However, this bonding was much weaker with AgF and CuF. Molecules MF₂ and MF₃ (M=Au, Ag, Cu) were identified from the isotopic distribution of the Cu and Ag atoms, comparison of the frequencies for three metal fluorides, and theoretical frequency calculations. The AuF₅ molecule was characterized by its strongest stretching mode and theoretical frequency calculations. Additional evidence was observed for the formation of the Au₂F₆ molecule.
Title
Matrix Infrared Spectroscopy and Quantum-Chemical Calculations for the Coinage-Metal Fluorides: Comparisons of Ar-AuF, Ne-AuF, and Molecules MF2 and MF3
Author
Xuefeng Wang, Lester Andrews, Felix Brosi, Sebastian Riedel
Date
2013
Source(s)
Appeared in
Chem. Eur. J. 2013, 19, 4, 1397–1409
Citation
DOI: 10.1002/chem.201203306
Language
eng