Polychloride Monoanions from [Cl3]− to [Cl9]−: A Raman Spectroscopic and Quantum Chemical Investigation
Robin Brückner, Heike Haller, Mathias Ellwanger, Sebastian Riedel – 2012
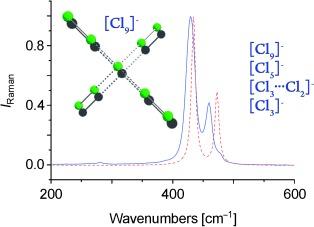
Polychloride monoanions stabilized by quaternary ammonium salts are investigated using Raman spectroscopy and state-of-the-art quantum-chemical calculations. A regular V-shaped pentachloride is characterized for the [N(Me)₄][Cl₅] salt, whereas a hockey-stick-like structure is tentatively assigned for [N(Et)₄][Cl₂⋅⋅⋅Cl₃⁻]. Increasing the size of the cation to the quaternary ammonium salts [NPr₄]⁺ and [NBu₄]⁺ leads to the formation of the [Cl₃]⁻ anion. The latter is found to be a pale yellow liquid at about 40 °C, whereas all the other compounds exist as powders. Further to these observations, the novel [Cl₉]⁻ anion is characterized by low-temperature Raman spectroscopy in conjunction with quantum-chemical calculations.
Title
Polychloride Monoanions from [Cl3]− to [Cl9]−: A Raman Spectroscopic and Quantum Chemical Investigation
Author
Robin Brückner, Heike Haller, Mathias Ellwanger, Sebastian Riedel
Date
2012
Source(s)
Appeared in
Chem. Eur. J. 2012, 18, 18, 5741-5747
Citation
DOI: 10.1002/chem.201103659
Language
eng