Investigation of Polybromide Monoanions of the Series [NAlk4][Br9] (Alk = Methyl, Ethyl, Propyl, Butyl)
Heike Haller, Mathias Ellwanger, Alexander Higelin, Sebastian Riedel – 2011
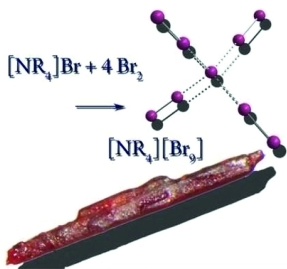
Polybromide monoanions of the series [NAlk₄][Br₉] (Alk = methyl, ethyl, propyl, butyl) have been investigated. The corresponding salts were characterized by Raman spectroscopy, single-crystal X-ray diffraction and state-of-the-art quantum-chemical calculations. Comparisons between their crystal structures and the gas-phase calculations have been made whereas the latter shows regular tetrahedral arrangement. Usually the nonabromides contain one central Br⁻ anion at which all other four Br₂ molecules are coordinated. The only exception is [NBu₄][Br₉] where also a [Br₃]⁻ unit is found.Quantum-chemical calculations show that dispersion as well as spin-orbit corrections alter the results only marginally and can be neglected. Conductivity measurements show a surprisingly high conductivity of these polybromide monoanions. It is found that the tetraalkylammonium nonabromide salts (alkyl = methyl, ethyl, propyl, and butyl) show decreasing conductivity as the size of the cation increases. Making thesepolybromide monoanions possible electrolytes in batteries or dye-sensitized solar cells.