Analysis of Tertiary Phosphanes, Arsanes, and Stibanes as Bridging Ligands in Dinuclear Group 9 Complexes
Sandra Schinzel, Robert Müller, Sebastian Riedel, Helmut Werner, Martin Kaupp – 2011
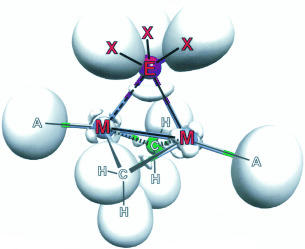
The unusual bridging and semi-bridging binding mode of tertiary phosphanes, arsanes, and stibanes in dinuclear low-valent Group 9 complexes have been studied by density functional methods and bonding analyses. The influence of various parameters (bridging and terminal ligands, metal atoms) on the structural preferences and bonding of dinuclear complexes of the general composition [A¹ M¹(μ-CH₂)₂(μ-EX₃)M² A²] (M¹, M²=Co, Rh, Ir; A¹, A²=F, Cl, Br, I, κ²-acac; E=P, As, Sb, X=H, F, CH₃) has been analyzed. A number of factors have been identified that favor bridging or semi-bridging modes for the phosphane ligands and their homologues. A more symmetrical position of the bridging ligand EX₃ is promoted by more polar EX bonding, but by less electronegative (softer) terminal anionic ligands. Among the Group 9 metal elements Co, Rh, and Ir, the computations clearly show that the 4d element rhodium exhibits the largest preference for a {M¹(μ-EX₃)M²} bridge, in agreement with experimental observation. Iridium complexes should be valid targets, whereas cobalt does not seem to support well a symmetric bridging mode. Analyses of the Electron Localization Function (ELF) indicate a competition between a delocalized three-center bridge bond and direct metal–metal bonding.