Quantum Chemical Study of Trivalent Group 12 Fluorides
Sebastian Riedel, Martin Kaupp, Pekka Pyykkö – 2008
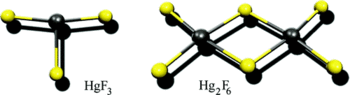
In view of the recent experimental observation of the existence of tetravalent Hgᴵⱽ in HgF₄, a quantum chemical study of various trivalent group 12 Mᴵᴵᴵ fluoride complexes has been carried out. The M-F bonds in neutral MF₃ are relatively weak, making these species unlikely targets. Some additional stability can be obtained by dimerizing HgF₃ to Hg₂F₆, which has a doubly fluorine-bridged triplet ground state. Anionic [MF₄]⁻ and [MF₅]²⁻ species are found to be more stable toward F2 elimination and M-F bond breaking than neutral MF₃.
Title
Quantum Chemical Study of Trivalent Group 12 Fluorides
Author
Sebastian Riedel, Martin Kaupp, Pekka Pyykkö
Date
2008
Source(s)
Appeared in
Inorg. Chem. 2008, 47, 8, 3379–3383
Citation
DOI: 10.1021/ic702384y
Language
eng