High-Valent Technetium Fluorides. Does TcF7 Exist?
Sebastian Riedel, Manuel Renz, Martin Kaupp – 2007
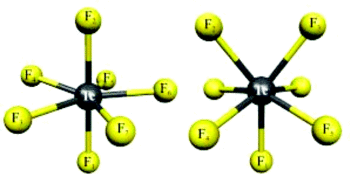
The structures and stabilities of technetium fluorides in high oxidation states have been studied quantum-chemically at density functional theory (B3LYP) and coupled-cluster (CCSD(T)) levels. The calculations indicate clearly that technetium heptafluoride, TcF₇, has a good chance of existence and preparation, thus providing the first heptafluoride in the 4d series. The [TcF₆]⁺ cation, a potential precursor, is also computed to be thermochemically stable against gas-phase elimination reactions. The problem with such highly fluorinated complexes appears thus to be mainly in difficult synthetic access under typical condensed-phase conditions. Matrix-isolation techniques or gas-phase experiments appear to be better suited as starting points.