After 20 Years, Theoretical Evidence That “AuF7” Is Actually AuF5·F2
Daniel Himmel, Sebastian Riedel – 2007
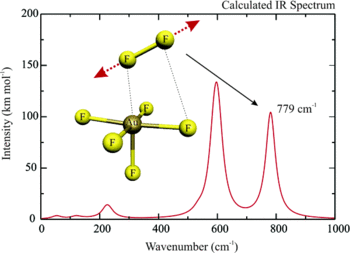
Quantum chemical calculations at DFT (BP86, B3LYP, BHLYP), MP2, CCSD, and CCSD(T) levels have been carried out on various fluoro complexes of gold in oxidation states +V through +VII, to evaluate the previously claimed existence of AuF₇. The calculations indicate clearly that elimination of F₂ from AuF₇ is a strongly exothermic reaction, with a low activation barrier. This is inconsistent with the reported stability of AuF₇ up to room temperature. A reported experimental vibrational frequency at 734 cm⁻¹ for AuF₇ could not be verified computationally. It is concluded that the reported observation of AuF₇ was probably erroneous. As the calculations indicate also an extremely large electron affinity and little stability for AuF₅, Au⁺ⱽ remains the highest well-established gold oxidation state.