Platinum fluorides beyond PtF6?
Sebastian Riedel – 2007
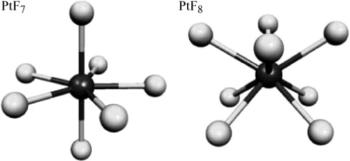
Quantum-chemical calculations at DFT B3LYP and ab initio MP2, CCSD, and CCSD(T) levels have been performed on various binary fluorides of platinum up to formal oxidation state +VIII, to evaluate the stability of these species. The calculations indicate clearly that elimination of F₂ from PtF₈ is a strongly exothermic reaction, with a moderate activation barrier. An exothermic decay is also observed for the homolytic bond breaking. Furthermore, our investigations suggest that both decomposition channels of PtF₇ are exothermic. The existence of platinum fluorides higher than PtF₆ is therefore highly unlikely.
Title
Platinum fluorides beyond PtF6?
Author
Sebastian Riedel
Date
2007
Source(s)
Appeared in
J. Fluorine Chem. 2007, 128, 8, 938 - 942
Citation
DOI: 10.1016/j.jfluchem.2007.04.001
Language
eng