Background
Synaptic plasticity, meaning changes in structure and/or function of the synaptic connections between neurons, is a cellular basis of learning and memory processes in the brain. Particularly, the formation of new, additional synapses within neuronal circuits is considered to be a primary mechanism for long-term synaptic plasticity. However, the elementary mechanisms controlling functional and structural assembly of synapses remain understood only very poorly. Aim of our laboratory is to study synaptic assembly and remodeling processes in intact living preparations, particularly concentrating on the relation between structural and functional organization.
Model system: Glutamatergic synapses easily accessible for genetics and imaging
Glutamate is the dominant excitatory neurotransmitter of our brain. Primary model of our group are glutamatergic synapses of the Drosophila neuromuscular junctions (NMJs). While being similar to glutamatergic CNS synapses of mammals in terms of ultrastructure and molecular composition, the NMJ combines a comparatively simple architecture (see figure) with straightforward genetic accessibility. In addition, our group recently also devised protocols to allow in vivo imaging of identified synapse populations over days of NMJs in living larvae, using confocal and two-photon-microscopy (Rasse et al., 2005). With this end, the protein dynamics organizing the synapse assembly process could be directly visualized in living animals, e.g. by using in vivo fluorescence recovery experiments. Our approaches are finally complemented by biochemical, ultrastructural and electrophysiological analyses.
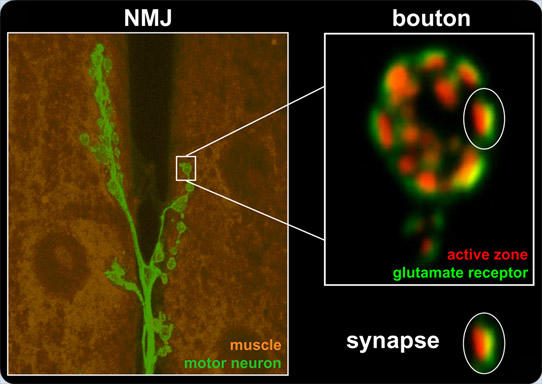
Morphological organization of the DrosophilaNeuromuscular Junction (NMJ)
Shown is a larval muscle (orange) innervated by motoneurons (green) branching into numerous boutons (left panel). Per bouton, 10-20 synapses consisting of postsynaptic glutamate receptor fields and associated presynaptic active zone (right panel) are found.
More information is available about our current projects and STED microscopy. Click here for a summary of our research.



