{μ‐PbSe}: A Heavy CO Homologue as an Unexpected Ligand
G. Thiele, Y. Franzke, F. Weigend, S. Dehnen – 2015
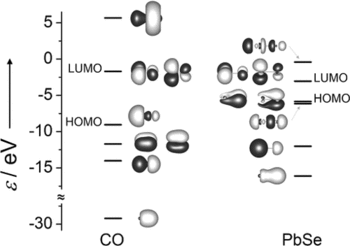
Reactions of [K(18‐crown‐6)]2[Pb2Se3] and [K([2.2.2]crypt)]2[Pb2Se3] with [Rh(PPh3)3Cl] in en (ethane‐1,2‐diamine) afforded ionic compounds with [Rh3(PPh3)6(μ3‐Se)2]− and [Rh3(CN)2(PPh3)4(μ3‐Se)2(μ‐PbSe)]3− anions, respectively. The latter contains a PbSe ligand, a rather uncommon homologue of CO that acts as a μ‐bridge between two Rh atoms. Quantum chemical calculations yield a significantly higher bond energy for PbSe than for CO, since the size of the ligand orbitals better matches the comparably rigid Rh‐Se‐Rh angles and the resulting Rh⋅⋅⋅Rh distance. To rationalize the bent coordination of the ligand, orbitals with significant ligand contributions and their dependence on the bonding angle were investigated in detail.
Titel
{μ‐PbSe}: A Heavy CO Homologue as an Unexpected Ligand
Verfasser
G. Thiele, Y. Franzke, F. Weigend, S. Dehnen
Datum
2015
Zitierweise
Angew. Chem. 2015, 127, 11437;
Angew. Chem. Int. Ed. 2015, 54, 11283.