On the Upper Limits of Oxidation States in Chemistry
Shu-Xian Hu, Wan-Lu Li, Jun-Bo Lu, Junwei Lucas Bao, Haoyu S. Yu, Donald G. Truhlar, John K. Gibson, Joaquim Marcalo, Mingfei Zhou, Sebastian Riedel, W.H.Eugen Schwarz, Jun Li – 2018
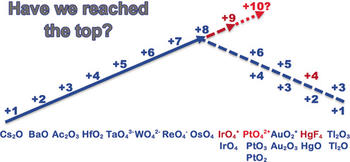
The concept of oxidation state (OS) is based on the concept of Lewis electron pairs, in which the bonding electrons are assigned to the more electronegative element. This approach is useful for keeping track of the electrons, predicting chemical trends, and guiding syntheses. Experimental and quantum‐chemical results reveal a limit near +8 for the highest OS in stable neutral chemical substances under ambient conditions. OS=+9 was observed for the isolated [IrO₄]⁺ cation in vacuum. The prediction of OS=+10 for isolated [PtO₄]²⁺ cations is confirmed computationally for low temperatures only, but hasn't yet been experimentally verified. For high OS species, oxidation of the ligands, for example, of O⁻² with formation of .O⁻¹ and O−O bonds, and partial reduction of the metal center may be favorable, possibly leading to non‐Lewis type structures.