Isolation and structural and electronic characterization of salts of the decamethylferrocene dication
M. Malischewski, M. Adelhardt, J. Sutter, K. Meyer, K. Seppelt – 2016
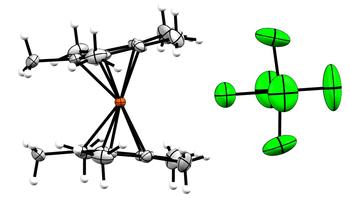
Ferrocene and its decamethyl derivative [Cp*₂Fe] are the most common standards for nonaqueous electrochemical investigations because of their well-defined and only mildly solvent-dependent reversible Fe(II)/Fe(III) redox couple. Higher oxidation states have only rarely been studied. We report the isolation and crystallographic and spectroscopic characterization of surprisingly stable Fe(IV) salts of the [Cp*₂Fe]²⁺ dication, produced by oxidation of [Cp*₂Fe] with AsF₅, SbF₅, or ReF₆ in neat sulfur dioxide as well as [XeF](Sb₂F₁₁) in neat hydrogen fluoride. The Sb₂F₁₁⁻ salt exhibits a metallocene with the expected mutually parallel arrangements of the Cp* rings, whereas the As₂F₁₁⁻, AsF₆⁻, SbF₆⁻, and ReF₆⁻ salts manifest tilt angles ranging from 4° to 17°. Both 57Fe Mössbauer spectroscopy and superconducting quantum interference device magnetization studies reveal identical d-orbital splitting with an S = 1, ³E ground state based on the 3d electronic configuration e₂g³a₁g¹ of all [Cp*₂Fe]²⁺ salts.