Research
Long non-coding RNAs, transposons, and DNA Repair in Plants
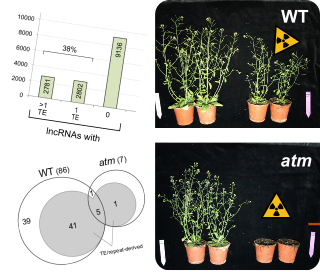
Plants are continuously exposed to DNA damaging radiation and other physical or chemical cytotoxic stresses because they are sessile and cannot actively escape exposure to such environmental stresses. As DNA damage like double-strand breaks induced by radiation is a severe threat to genome integrity, plants have evolved highly efficient DNA repair mechanisms. The induction of chromosome breaks in Arabidopsis thaliana by exposure to X-rays results not only in transcriptional activation of more than 1400 protein-coding genes but also in the activation of long non-coding RNAs (lncRNAs) (Wang et al., 2016), which frequently overlap with or contain transposon sequences. Several lncRNAs of unknown function are massively upregulated and this transcriptional DNA damage response is dependent on the DNA double-strand break-sensing protein kinase ATM (ataxia telangiectasia mutated), suggesting that these lncRNAs are involved in the regulation of cellular DNA damage repair processes. By mutant and molecular analyses we investigate the biological function of these DNA damage-responsive lncRNAs.
Priming of defense against herbivores feeding upon Arabidopsis thaliana
This research is a joint project of the R. Kunze and M. Hilker groups and part of the Collaborative Research Centre CRC973 "Priming and Memory of Organismic Responses to Stress".
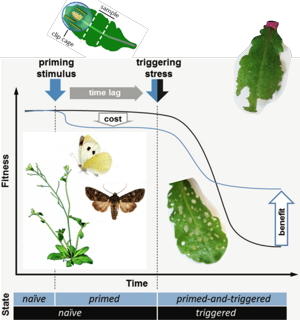
When herbivorous insects deposit eggs on a leaf, the larvae that hatch some days later will start feeding on these leaves and damage the plant. If plants can recognize egg deposits on their leaves, these can be reliable warning cues of subsequent larval herbivory and the plants get a chance to "prepare" for improved defense actions against the larvae. We found that egg deposition by the cabbage white butterfly (Pieris brassicae) onto Arabidopsis thaliana leaves, but not the exposure of the plant to the other stresses like 'chilling' or fungal infection, improves the plant´s anti-herbivore defense against larvae (Firtzlaff et al., 2016). We investigate the characteristics of the egg-associated stimulus that warns Arabidopsis of future herbivory, the function of Arabidopsis genes with altered transcriptional profiles in feeding damaged leaves that previously carried eggs, the molecular 'memory' of the plants for prior egg deposition, and the impact of the plant´s longevity on its primability and persistence of the primed state.