Multivalent Pseudorotaxanes: Cooperative Binding and Synthesis of Multiply Interlocked Rotaxanes
Multivalent binding plays a crucial role in many biochemical processes, for example virus docking to a cell. To understand multivalency, cooperative binding and the role of the nature of the spacer connecting the binding sites, di- and trivalent pseudorotaxanes are investigated. As synthetic molecules can be adjusted and designed with respect to many parameters, a systematic investigation is possible.
The Schalley group uses crown ether/secondary ammonium as well as amide pseudorotaxanes for these studies. Detailed analysis of the thermochemistry by applying the double mutant cycle concept allows determining chelate cooperativity and thus provides a more detailed understanding of the underlying binding processes.
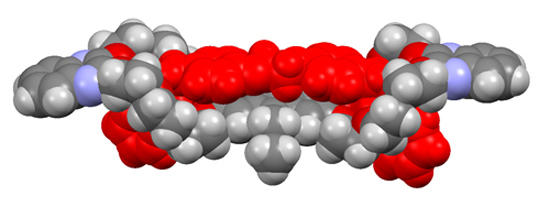
Crystal structure of a divalent crown/ammonium pseudorotaxane displaying positively cooperative binding due to favorable spacer-spacer interactions
Related Publications
-
189
Multivalency in the Gas Phase: H/D-Exchange Reactions Unravel the Dynamic 'Rock 'n Roll' Motion in Dendrimer-Dendrimer Complexes
Z. Qi, C. Schlaich, C.A. Schalley
Chem. Eur. J. 2013, 19, 14867-14875
-
177
Multivalency as a Chemical Organization and Action Principle
C. Fasting, C.A. Schalley, M. Weber, O. Seitz, S. Hecht, B. Koksch, J. Dernedde, C. Graf, E.-W. Knapp, R. Haag
Angew. Chem., 2012, 124, 10622-10650; Angew. Chem. Int Ed., 2012, 51, 10472-10498
-
164
Phenanthroline- and Terpyridine-Substituted Tetralactam Macrocycles: A Facile Route to Rigid Di- and Trivalent Receptors and Interlocked Molecules
E. V. Dzyuba, B. Baytekin, D. Sattler, C.A. Schalley
Eur. J. Org. Chem. 2012, 1171-1178
-
166
Synthesis of Multivalent Host and Guest Molecules for the Construction of Multithreaded Diamide Pseudorotaxanes
N.L. Löw, E.V. Dzyuba, B. Brusilowskij, L. Kaufmann, E. Franzmann, W. Maison, E. Brandt, D. Aicher, A. Wiehe, C.A. Schalley
Beilstein J. Org. Chem., 2012, 8, 234-245
-
163
Chelate Cooperativity and Spacer Length Effects on the Assembly Thermodynamics and Kinetics of Divalent Pseudorotaxanes
W. Jiang, K. Nowosinski, N.L. Löw, E.V. Dzyuba, F. Klautzsch, A. Schäfer, J. Huuskonen, K. Rissanen, C.A. Schalley
J. Am. Chem. Soc. 2012, 134, 1860-1868
-
140
Monitoring Self-Sorting by Electrospray Ionization Mass Spectrometry: Formation Intermediates and Error-Correction during the Self-Assembly of Multiply Threaded Pseudorotaxanes
W. Jiang, P.C. Mohr, A. Schäfer, C.A. Schalley
J. Am. Chem. Soc. 2010, 132, 2309-2320
-
124
Integrative Self-Sorting Is A Programming Language for High Level Self-Assembly
W. Jiang, C.A. Schalley
Proc. Natl. Acad. Sci. USA 2009, 106, 10425-10429


