Can Zinc Really Exist in Its Oxidation State +III?
Tobias Schlöder, Martin Kaupp, Sebastian Riedel – 2012
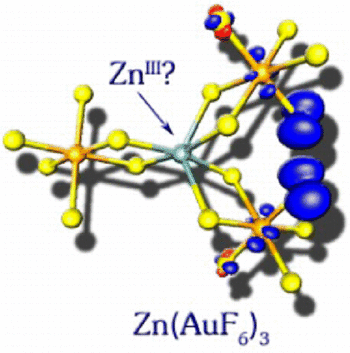
Very recently, a thermochemically stable Znᴵᴵᴵcomplex has been predicted by Samanta and Jena ( J. Am. Chem. Soc. 2012, 134, 8400−8403). In contrast to their conclusions we show here by quantum chemical calculations that (a) Zn(AuF₆)₃ is not a thermochemically feasible compound, and (b) even if it could be made, it would not represent a Znᴵᴵᴵ oxidation state by any valid definition.
Title
Can Zinc Really Exist in Its Oxidation State +III?
Author
Tobias Schlöder, Martin Kaupp, Sebastian Riedel
Date
2012
Source(s)
Appeared in
J. Am. Chem. Soc. 2012, 134, 29, 11977–11979
Citation
DOI: 10.1021/ja3052409
Language
eng